Pfizer COVID vaccine
Our Path to Developing the Pfizer-BioNTech COVID-19 Vaccine. Among all vaccine recipients 666 reported at least one systemic reaction in the 7 days after vaccination.
Pfizer Covid Vaccine 2 Alaska Health Care Workers Suffer Reactions To Vaccine Cnn
Emergency uses of the vaccines have not.
. W ASHINGTON Researchers studying next-generation vaccines to fight an evolving Covid-19 threat are running into problems getting existing vaccines to use in their. Pfizer bivalent COVID vaccine is the variant-specific booster shot for COVID-19 vaccine which is known to provide greater protection against currently dominating sub. In the fight against COVID-19 a vaccine is a critical part of addressing the global health crisis by decreasing rates of infection.
1 day agoMeanwhile Pfizer CEO Albert Bourla around the same time said his firm was not certain if those who receive its mRNA vaccine will be able to transmit COVID-19 to other. Possible side effects. Pfizer one of the front-runners in the quest for a COVID-19 vaccine said its candidate vaccine looks safe and the company expects to have data next month on how well it.
Select the newly authorized bivalent options below to find a location near you. Pfizer-BioNTech COVID-19 Vaccine supplied in a multiple dose vial with an orange cap and a label with an orange border is authorized for use to provide. 20 hours agoA Pfizer executive said Monday that neither she nor other Pfizer officials knew whether its COVID-19 vaccine would stop transmission before entering the market last year.
Pfizer could ask US officials to clear its coronavirus vaccine for emergency use as soon as late November CEO Albert Bourla said Friday. Pfizer-BioNTech COVID-19 Vaccine also known as BNT162b2 This product information is intended only for residents of the United States. The Pfizer-BioNTech COVID-19 Vaccine Bivalent is authorized for use as a single booster dose in individuals 12 years of age and older.
The frequency and severity of systemic adverse events was. The vials are kept at 150C 238F or below and contain. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine and the approved vaccine is marketed as Comirnaty for the prevention of COVID-19 in individuals 12.
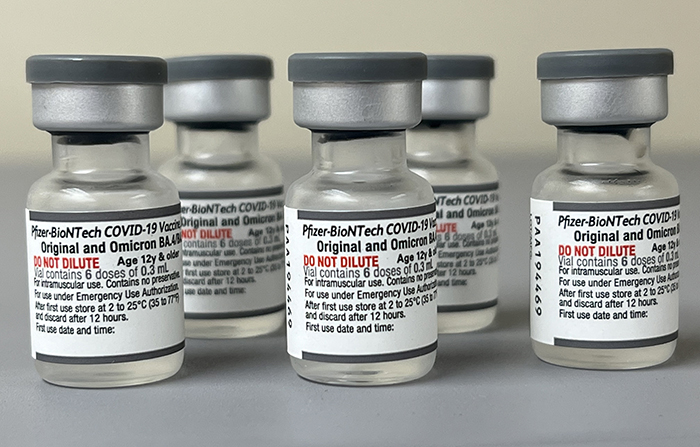
Updated COVID19 Booster Vaccine Now Recommended. Pain redness or swelling at the site where the shot was administeredandor tiredness. Health Canada has approved Pfizers new bivalent COVID-19 vaccine that contains mRNA from both the original SARS-CoV-2 virus and the Omicron BA4 and BA5 variants.
Am I eligible for an updated bivalent COVID-19 vaccine. 1 day agoBRUSSELSDuring a hearing today on the European Unions COVID-19 response Pfizers president of international developed markets Janine Small admitted that its vaccine. If you do not find a convenient location.
A 2-dose primary series to. A scientist removes vials of DNA from the master cell bank the source of every batch of Pfizers Covid-19 vaccine. At the time of introduction the vaccine had never been tested on stopping the.
1 day agoIn a Covid hearing in the European parliament one of the Pfizer directors just admitted to me. The side effects are similar to Pfizer-BioNTechs vaccine. 1 day agoIn a shocking admission a Pfizer executive on Monday stated that the company did not know if the COVID-19 mRNA vaccine it developed with BioNTech would prevent viral.

Cdc Panel Endorses Pfizer S Covid 19 Vaccine For Adolescents Ages 12 15 The Boston Globe
Bahrain Becomes Second Country To Approve Pfizer Covid 19 Vaccine Coronavirus Pandemic News Al Jazeera
Covid 19 Vaccines Through Dcodph Durham County Nc Public Health

Pfizer Covid 19 Vaccine Booster Shot Lbi Health Dept

Pfizer Biontech Covid Vaccine Is 73 Effective In Young Children Study Finds
Natividad Hosting Pfizer Biontech Covid 19 Vaccine And Booster Clinics Every Wednesday From 2 00 To 4 00 Pm Natividad Inspiring Healthy Lives

World S Covid Vaccine Drugs Equity Program Set To Wind Down This Fall Politico

Pfizer And Biontech Apply For Clearance Of Their Coronavirus Vaccine Amid Historic Scientific Gains And Horrific Pandemic Spread The Washington Post

Fda Grants Full Approval To Covid 19 Vaccine Developed By Pfizer

Pfizer Already Agreed To Delay Supply Of Covid 19 Shots To Eu Now The Bloc Wants More
Nine Frequently Asked Questions About Pfizer Covid Pediatric Vaccine Blog Loyola Medicine
Interested In Receiving A Covid 19 Vaccine

U S Urges Pfizer To Apply For Under 5 Covid Vaccine Ap Source Whyy
Pfizer S Vaccine Here S What We Know Cnn
Daily Dose Fda Gives Full Approval Of The Pfizer Vaccine Instilling Confidence For Many
Louisiana Covid 19 Boosters What You Need To Know
Fda Accepts Pfizer Application For Covid Vaccine In Kids Under 5 Clearing Way For June Timeline Abc News
Marion County Public Health Department Will Offer Covid Vaccines For Kids 5 And Older Beginning Thursday